Mine Koprulu leads study that identifies proteogenomic data which could result in new treatments for diseases such as diabetes
We systematically linked genetic variation, blood protein levels and disease risks to be able to differentiate proteins which are likely to cause a disease from those which may only be a result of diseases. Identifying causal proteins is important because only interventions on causal proteins will lead to safe and effective treatments.
Mine Koprulu
New research led by a Gates Cambridge Scholar has identified hundreds of proteins that might contribute to the onset of common, chronic metabolic diseases such as type 2 diabetes, and consequently pathways to potential treatments.
The study, published in Nature Metabolism, was led by an international research team from the Medical Research Council (MRC) Epidemiology Unit, University of Cambridge, the Precision Healthcare University Research Institute (PHURI) at Queen Mary University of London, and the Berlin Institute of Health at Charité (BIH) at Universitätsmedizin Berlin in Germany.
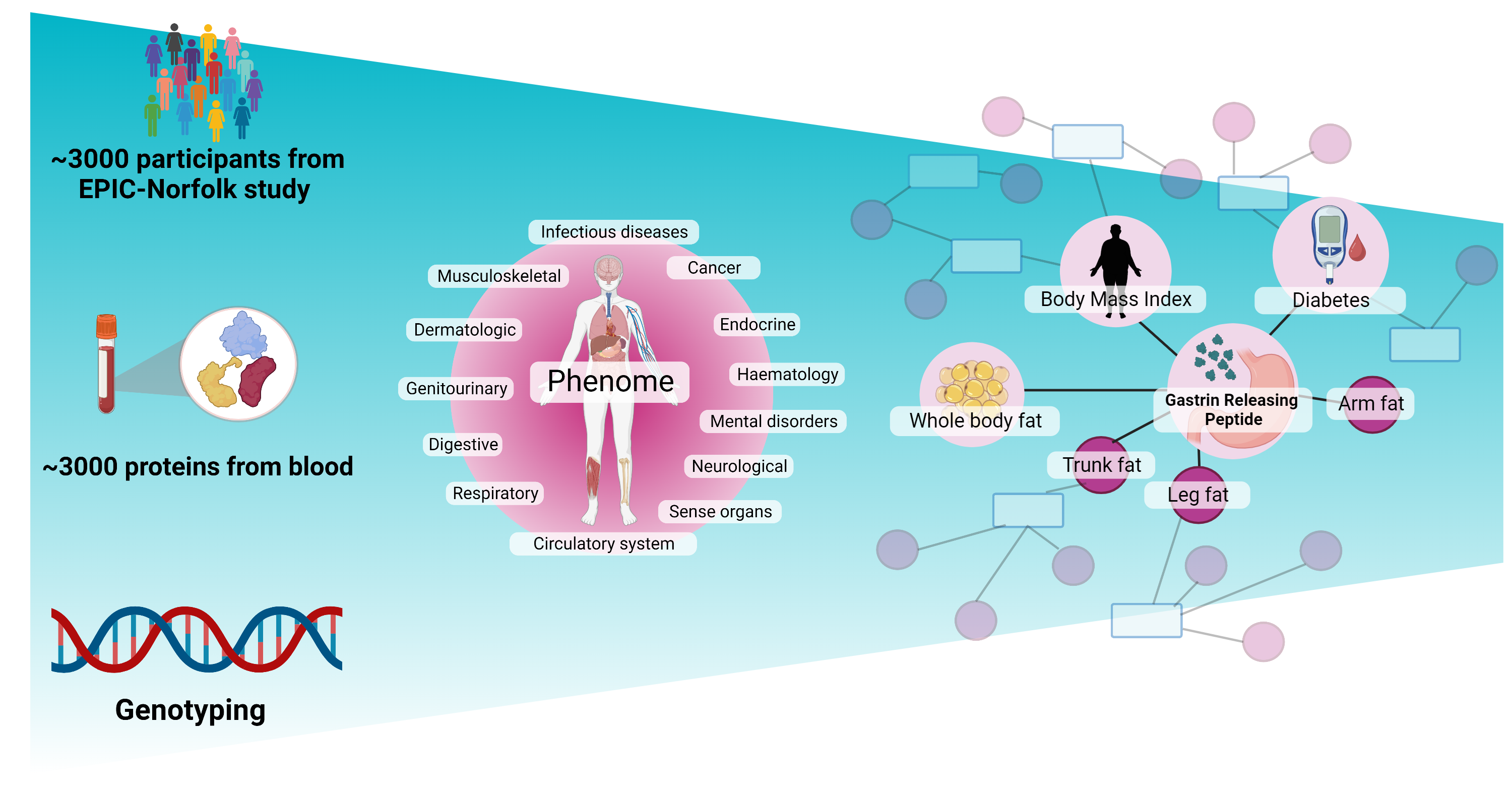
The researchers successfully linked more than 900 regions in the human genome to almost 3,000 proteins in our blood, with many of these not previously identified. The team then applied these findings to existing genetic studies for hundreds of diseases and found more than 500 gene-protein-disease links.
For example, the team showed for the first time that people with high levels of a hormone called GRP are less likely to develop type 2 diabetes, most likely because it decreases the chances of becoming overweight. This ‘proteogenomic’ evidence supports GRP as a potential target for the prevention and/or treatment of diabetes.
The study has led to a greater scientific understanding of hundreds of genome regions, which open the door to more targeted and ultimately successful treatment options in the future as proteins are essential functional units of the human body and the most common target of drugs that exist today.
Senior author Professor Claudia Langenberg, director of the Precision Healthcare University Research Institute (PHURI) at Queen Mary University of London and MRC Investigator and programme lead at the MRC Epidemiology Unit at the time of the study, said: “Thousands of regions in our genome have been identified to increase our risk for developing different diseases, but for most of them we have a poor understanding of why this is.
“By measuring and integrating information on thousands of proteins in human plasma, we were able to create robust links between the genes that encode these proteins to many different diseases and demystify about 200 regions. This really narrows down the potential therapeutic targets at each genomic region, often a bottleneck for translation of genomic discoveries.”
Senior author Professor Maik Pietzner, Professor at the PHURI and group leader at the Berlin Institute of Health at Charité (BIH), said “In an additional promising example, we identified a protein, called DKKL1, to be involved in multiple sclerosis that reinforces the depletion of certain immune cells – B-cells – as intervention.
“These early-stage results are exciting and show the potential of such technologies for drug discovery, not just for metabolic diseases.”
Mine Koprulu [2017], lead author of the study, Gates Cambridge scholar and PhD student at the MRC Epidemiology Unit, said: “The biological mechanisms underlying diseases are not always very well understood. To address this issue, we systematically linked genetic variation, blood protein levels and disease risks in this study to be able to differentiate proteins which are likely to cause a disease, for example diabetes, from those which may only be a result of diseases. Identifying causal proteins is important because only interventions on causal proteins will lead to safe and effective treatments. We are hugely grateful to the EPIC Norfolk volunteers and team who made this research possible.”











